Another is to make sure there is a sound product development plan (PDP) in place from the start. It is a multidisciplinary effort that requires extensive planning and coordination, as well as a clear vision of the end goal.Īs we saw previously, partnering with more experienced industry players who have been there before and know what to expect is one way that smaller biotech companies can improve their chances of success. 
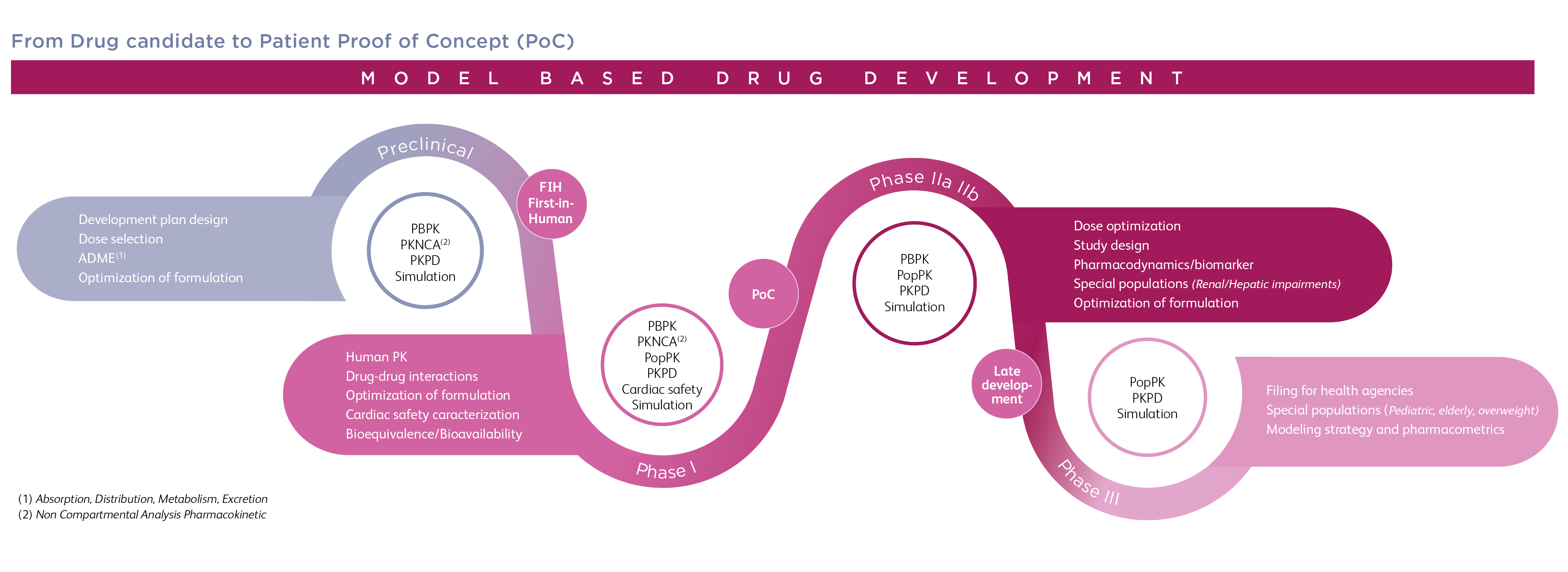
For the best chance of success, it is imperative to start out with a realistic understanding of what lies ahead, and a solid plan of how to arrive at an end product that is not only marketable but will give a healthy return on investment.Įven with sound science and technology as the starting point, turning a lead candidate into a commercially viable drug is no easy feat. The PDP: Where Opportunity Meets Realityīringing a beneficial new drug or therapy to patients is an exciting opportunity and a worthy goal, but not without significant risks. For each stage of the development process, the PDP clarifies the major goals and critical success factors, specifying how success will be measured and what needs to be done to mitigate any risks.Ī well-designed PDP not only increases the chances of success, it also plays an important role in helping the program teams reduce cost of goods, maximize efficiency and shorten time to market. It serves as a step-by-step guide to arrive at the envisioned drug product. The PDP is a strategic document that creates a detailed and comprehensive picture of the development strategy. What is a Product Development Plan (PDP)? In this article, we explore why and how to use this pivotal tool to formulate a winning drug development strategy. How do investors and executive managers bridge the gap between identifying a promising drug candidate and turning out a commercially successful product? While there is no simple answer to this question, one thing is certain: behind every successful new drug, there is a well-crafted product development plan (PDP). Yet for every success, there are countless tales of defeat. In 2020, 63% of approved new therapeutic drugs in Europe and the US came from small-to-medium enterprises. Without doubt, biotech startups and smaller pharmaceutical companies have become the major drivers of innovation in the pharmaceutical industry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
